
Be is exception to octet rule 2 180 Linear 3. PCl5 5 7 x 5 40 40 10 30 0 5 120.
Hint in this worksheet as in all chemistry problems youll see.
Vsepr worksheet with answers. Molecular Shape and VSEPR Theory Molecule Total valence electrons Lewis Structure Steric Number Electron Group Geometry Molecular Geometry Hybridization Ex. H2O 8 4 Tetrahedral Bent CO2 G-NH3 5-3 BF3. CH3Cl SiF5 eII ClF3 T Answer key 4 066-3 CO.
2 linear linear sp N-7-3 µ a tetrahedral Trpicpgoanmialdae sp suis B3. Lewis structure use rules for drawing Lewis structures C. Build molecule see pg 247 sketch give bond angles D.
Number of peripheral atoms E. Bond angles list all F. CH4 4 1095 Tetrahedral 2.
BeF2 2 7 x 2 16 16 4 12 0 Note. Be is exception to octet rule 2 180 Linear 3. PCl5 5 7 x 5 40 40 10 30 0 5 120.
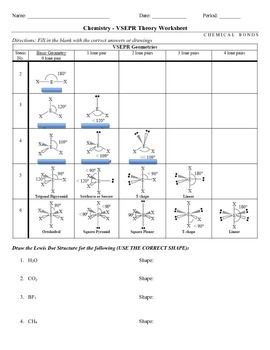
VSEPR Worksheet W 318 Everett Community College Tutoring Center Student Support Services Program 1 Briefly describe the primary ideas behind VSEPR theory. 2 For each of the following compounds a Lewis structure determine the bond angles and molecular shapes for all atoms. A BI 3 b CH 4 c NF 3 d C 2 H 2.
1 Electrons are negatively charged and like repels like so the atoms in a. Worksheet 13 - Molecular Shapes The shapes of molecules can be predicted from their Lewis structures by using the VSEPR Valence Shell Electron Pair Repulsion model which states that electron pairs around a central atoms will assume a geometry that keeps them as far apart from each other as possible. This is illustrated by the drawings below.
Lewis Structures VSEPR Polarity IM Forces - Answers For each of the following molecules draw the Lewis structure with any resonance structures if applicable indicate the molecular shapes and bond angles indicate the molecular polarity if any and identify the major intermolecular force in each compound. Hint in this worksheet as in. Worksheet 13 - Molecular Shapes The shapes of molecules can be predicted from their Lewis structures by using the VSEPR Valence Shell Electron Pair Repulsion model which states that electron pairs around a central atoms will assume a geometry that keeps them as far apart from each other as possible.
This is illustrated by the drawings below. VSEPR Theory Now that we have an understanding of covalent bonding and how atoms share electrons to form molecules and polyatomic ions we will use Lewis dot structures to predict electronic and molecular geometries. In turn the geometries of the molecules will influence many different physical and chemical properties like melting point boiling point surface tension viscosity.
Answer the following questions and check your answers below. These problems are for practice only will not be graded. Be sure you know how to draw correct Lewis Dot Structures and are able to correctly predict the electronic arrangement and molecular geometry before going on to the lab assignment.
Remember formal charges can sometimes help you decide which is the best Lewis Dot Structure. Some of the worksheets below are Molecular Geometry Worksheet with Answers exercises like draw Lewis structures and determine the molecular geometry and polarity for each of the molecules below. Find out how can molecular shapes be predicted using the VSEPR theory and an activity to help you learn learn how to predict molecular shapes.
Once you find your worksheets you. Vsepr worksheet with answers fresh covalent worksheet of responses to a table with molecular geometry source. Related searches for molecular geometry answers molecular geometry response keymolecular geometry response key pdfmolecular geometry response.
Geometry is linear 7 one s and two pbitals hybridize to form sp2 trigonal plantar. Some of the worksheets for this concept are read. We found some Images about Vsepr Theory Worksheet With Answers.
VSEPR - Chemistry LibreTexts table 1 001jpg. Chem 11 Lewis Structures and VSEPR Mr. Sius Chemistry Classes.
AP Chemistry 2014-2015 - Mrs Grindles Science Site Attachments. Geometry Worksheet Answers Free Worksheets Library Download and. Worksheet 15 - Molecular Shapes The shapes of molecules can be predicted from their Lewis structures by using the VSEPR Valence Shell Electron Pair Repulsion model which states that electron pairs around a central atoms will assume a geometry that keeps them as far apart from each other as possible.
This is illustrated by the drawings below. VSEPR worksheets answers Self-test Chapter 10 Authors version of a practice test Following the example fill in the 2D and 3D structures and information associated with each compound and ion. Vsepr Worksheet with Answers.
The shapes of molecules can be Self-test Chapter 10 Answers Writing Lewis Topic 9 Read PDF Vsepr Theory Practice With Answers Problems VSEPR practice problems. Lewis Structures VSEPR Polarity IM Forces - Answers For each of the following molecules draw the Lewis structure with any resonance structures if applicable indicate the molecular shapes and bond angles indicate the molecular polarity if any and identify the major intermolecular force in each compound. Hint in this worksheet as in all chemistry problems youll see.
Vsepr Worksheet With Answers Eventually you will very discover a further experience and completion by spending more cash. Pull off you agree to that you require to get those all needs similar to having significantly cash. Why dont you try to acquire something basic in the beginning.
Thats something that will guide you to comprehend even more in relation to the globe. Lewis and VSEPR worksheet answers. CS 2 a total valence electrons carbon 2sulfur 4 26 16 b the Lewis dot structure will have the carbon as the central atom.
C Thus the electronic shape is linear because there are two regions of electron density. D The molecular shape is also linear since there are no lone pairs on the central atom. NF 3 a the total valence.
Shape And Structure Of Molecules Vsepr Worksheet With Answers When matter is in a solid state its molecules are packed close together so they can t move around this creates a stable structure with a definite size and shape teach your child about this first C all the p cl bonds in pcl 5 molecules standing answer a i ibr 2 it has 2 bond pairs and 3 lone pairs therefore according to vsepr theory. Vsepr worksheet with answers pdf The Lewis and VSEPR spreadsheets correspond to 1 January 2007. CS2a total number of valensic electrons carbon 2sulphur 4 26 16b In Lewis sulphur structure carbon is the middle atom.
C Therefore the electronic form is linear because there are two areas of electron density. D The molecular form is also linear since the middle atom does not. Sep 23 2020 - Vsepr Worksheet with Answers.
32 Vsepr Worksheet with Answers. 66 Tutorial atomic Structure 4 Answer Key with Video and Pdf. Answers Vsepr Worksheet With Answers Thank you very much for downloading vsepr worksheet with answers.
As you may know people have look numerous times for their chosen books like this vsepr worksheet with answers but end Page 19. Read Online Vsepr Worksheet With Answersup in malicious downloads. Rather than enjoying a good book with a cup of tea in the afternoon instead.